Detergents and dispersants work together to break down dirt, oils, and mineral buildup. Surfactants reduce water’s surface tension and form micelles that trap oil and dirt so they can be rinsed away. Dispersants bind with minerals in hard water, preventing deposits and enabling better cleaning. Enzymes and bleach boost stain removal and brighten fabrics. Understanding how these components work helps you get the most effective and eco-friendly cleaning results. Keep exploring to learn more tips and tricks.
Key Takeaways
- Detergents contain surfactants that reduce water surface tension, aiding in spreading and penetrating surfaces for effective cleaning.
- Surfactants form micelles that trap oils and dirt, making them easier to rinse away from surfaces and fabrics.
- Dispersants bind to minerals like calcium and magnesium in hard water, preventing mineral deposits and soap scum formation.
- Detergents often include enzymes and bleach to break down stains and remove stubborn dirt effectively.
- Both detergents and dispersants enhance cleaning efficiency, protect appliances, and promote environmental sustainability through specialized formulations.

Enzyme Laundry Booster Odor Remover – 2 lbs Unscented Enzymatic Clothes Stain Cleaner Powder, Natural Deodorizer with Bio Active Enzymes, Detergent Additive Eliminator for Sweat, Oil, Blood – 64 Loads
Powerful Bio Enzymatic Cleaner: ACTIVE Laundry Odor Removers Boost your normal laundry detergent to effectively target Stains &…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
How Do Surfactants Help Clean Surfaces?

Surfactants help clean surfaces by reducing the surface tension of water, allowing it to spread more easily and penetrate fabrics or surfaces better. When you use a detergent, surfactants surround dirt, grease, and oil particles with their unique structure—hydrophobic tails and hydrophilic heads. The tails latch onto oily substances, breaking them apart, while the heads face outward toward the water. This action lifts the dirt from the surface, making it easier to rinse away. Surfactants also facilitate emulsification, dispersing oil and grease throughout the wash water so they don’t reattach to your clothes or surfaces. By lowering water’s surface tension, surfactants enable water to reach into tiny fibers and crevices, ensuring a more thorough clean. This process is essential for effective removal of stubborn dirt and grime. Understanding surface tension is key to appreciating how detergents work to achieve cleanliness.

Rockin' Green Hard Rock Travel Laundry Detergent Sheets for Hard Water – Mess-Free Laundry Sheet Detergent with Sodium Bicarbonate – Easily Dissolvable Laundry Soap Sheets (104 Loads, Unscented)
Formulated for Hard Water – Hard Rock Rockin' Green laundry detergent sheets fight tough stains and banish odors…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
What Are Micelles and How Do They Remove Oil and Dirt?

When surfactants surround dirt and oil particles, they form structures called micelles that play a key role in cleaning. These micelles have hydrophobic tails inward, trapping oils and grease, while their hydrophilic heads face outward, interacting with water. This arrangement allows the micelles to encapsulate oily contaminants, effectively lifting them away from surfaces. As you rinse, the water washes away the micelles along with the trapped dirt and oil, leaving the surface clean. Micelles also help emulsify oil, dispersing tiny droplets throughout the wash water, preventing reattachment. This process ensures that stubborn oils and dirt are suspended and easily rinsed away, making your cleaning more efficient. Understanding micelles reveals how detergents turn oily grime into manageable particles for effective removal. Additionally, the formation of micelles is influenced by the surfactant concentration, which determines how effectively oils are encapsulated and removed.

BLUELAND 100% Plastic-Free Dishwasher Detergent Tablets, 60 Count, Free & Clear, Eco-Friendly Dishwashing Pods, Natural, USDA Certified biobased
PLASTIC-FREE, ECO FRIENDLY DISHWASHER DETERGENT TABLETS. Hardworking without the harsh chemicals, our NEW and improved, eco-friendly Dishwasher Detergent…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
What Types of Surfactants Are Used in Detergents?

You’ll find different types of surfactants in detergents, each designed for specific cleaning needs. Anionic surfactants, like alkyl sulfates, are common because they effectively remove dirt and grease. Understanding how these surfactants work helps you choose the right detergent for your cleaning tasks.
Types of Surfactants Used
What types of surfactants are commonly used in detergents? You’ll typically find anionic, non-ionic, and cationic surfactants. Anionic surfactants, like alkyl sulfates and sulfonates, are the most common and excel at removing dirt and grease due to their negative charge. Non-ionic surfactants, such as alcohol ethoxylates, don’t carry a charge and work well in hard water, providing good cleaning without forming soap scum. Cationic surfactants, usually quaternary ammonium compounds, act as disinfectants and fabric softeners but aren’t used for heavy cleaning. Each type serves a specific purpose, and many detergents blend them to optimize cleaning, foaming, and fabric care. These variations allow formulators to create versatile products tailored to different cleaning needs, often involving specific surfactant properties that enhance their effectiveness.
Function of Anionic Surfactants
Anionic surfactants play a crucial role in detergents because of their strong ability to remove dirt and grease. They work by forming micelles that trap oily particles, allowing water to wash them away efficiently. These surfactants have negatively charged heads that interact with water and hydrophobic tails that attach to oils. Common types include alkyl sulfates, sulfonates, and carboxylates. Their effectiveness depends on their structure and interaction with soil and water conditions. Here’s a quick overview: Surface activity influences how well they perform under different washing environments.
| Type | Common Examples | Key Characteristics |
|---|---|---|
| Alkyl Sulfates | Sodium lauryl sulfate | Good foaming, strong cleaning |
| Sulfonates | Linear alkylbenzene sulfonate | Cost-effective, versatile |
| Carboxylates | Soap salts, fatty acids | Mild, biodegradable |
| Non-ionic | Alcohol ethoxylates | Reduced foam, effective in hard water |

Chemical Guys Foaming Citrus Fabric Clean Carpet & Upholstery Cleaner – Car Carpet Cleaner Spray for Seats and Floor Mats, Removes Stains &Odors, Safe for Cars, Home and Office, 16 oz Citrus Scent
Powerful Cleaning (CWS21916SW) – Ready-to-use Chemical Guys fabric cleaner spray breaks down dirt, grime, and stains from carpets,…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
How Do Enzymes and Bleach Boost Cleaning Power?

Enzymes and bleach considerably enhance cleaning by targeting specific stains and soils. Enzymes break down proteins, fats, and carbohydrates, making dirt easier to remove, while bleach oxidizes stubborn stains for a brighter wash. Proper gear care also helps maintain the effectiveness of your cleaning products over time. Together, they boost your detergent’s effectiveness, especially on tough spots.
Enzyme Action Mechanisms
Enzymes play a essential role in boosting cleaning power by specifically targeting and breaking down common stains and dirt components. They do this by catalyzing the breakdown of proteins, lipids, and polysaccharides into smaller, easier-to-remove molecules. This process makes stains less stubborn and easier to rinse away. Here are how enzymes improve cleaning:
- Target Specific Molecules: Enzymes recognize and bind to particular stain components like proteins or fats.
- Catalyze Breakdown: They accelerate chemical reactions that split large molecules into smaller fragments.
- Enhance Detergency: Smaller molecules are more soluble and easier to wash away with water.
- Work at Lower Temperatures: Enzymes are effective even in cold water, saving energy and preserving fabrics.
This targeted action considerably boosts overall cleaning efficiency.
Bleach Stain Removal
Bleach and enzymes work together to markedly boost stain removal by attacking different types of dirt and discoloration. Bleach, usually containing hydrogen peroxide or sodium hypochlorite, oxidizes color molecules, breaking down stubborn stains like wine, coffee, or ink. Enzymes, on the other hand, target organic matter such as proteins, fats, and carbohydrates, breaking them into smaller, washable fragments. When combined, bleach quickly whitens and brightens fabrics, while enzymes dissolve organic residues that bleach alone can’t handle. This synergy results in more effective cleaning, especially for tough stains. You’ll notice improved fabric whiteness and stain removal, even in cold water. Just remember, using the right amount and proper washing conditions helps maximize their power without damaging your clothes. Being aware of ethical considerations around chemical use can help consumers make more responsible choices when selecting cleaning products.
How Does Water Hardness Affect Detergent Performance?

Water hardness considerably impacts detergent performance because the calcium and magnesium ions in hard water react with surfactants to form insoluble scum. This scum reduces the surface activity of detergents, making them less effective at lifting dirt and oils. To counteract this, you can:
Hard water minerals diminish detergent effectiveness by forming insoluble scum that hampers cleaning.
- Use chelating agents that bind calcium and magnesium ions.
- Add water softeners to replace mineral ions with sodium or potassium.
- Increase detergent dosage to compensate for mineral interference.
- Opt for detergents formulated for hard water, often containing built-in chelators.
- Incorporate foam rolling routines to help improve overall recovery and HRV, ensuring your body is better prepared for effective cleaning and maintenance.
Without these measures, mineral deposits can also damage washing machines and pipes over time, further hindering cleaning efficiency. Understanding water hardness helps you choose the right products and techniques for ideal cleaning results.
What Factors Influence How Well a Detergent Works?

Several factors directly impact how effectively a detergent cleans your clothes. First, pH level plays a pivotal role; a pH between 7 and 11 optimizes surfactant activity and enzyme performance. Second, water temperature matters—hot water enhances dirt removal, but cold water with specialized enzymes can also be effective. Third, agitation helps loosen dirt particles, making them easier to wash away. The amount of detergent used is essential—using too little leaves stains, while too much can cause soap scum buildup. Additionally, the formulation matters: surfactant type, presence of enzymes, bleaching agents, and water softeners all influence cleaning power. Finally, fabric type and soil level affect how well the detergent works; heavier soils and delicate fabrics require tailored solutions for best results.
How Do Dispersants Make Cleaning Better in Hard Water?
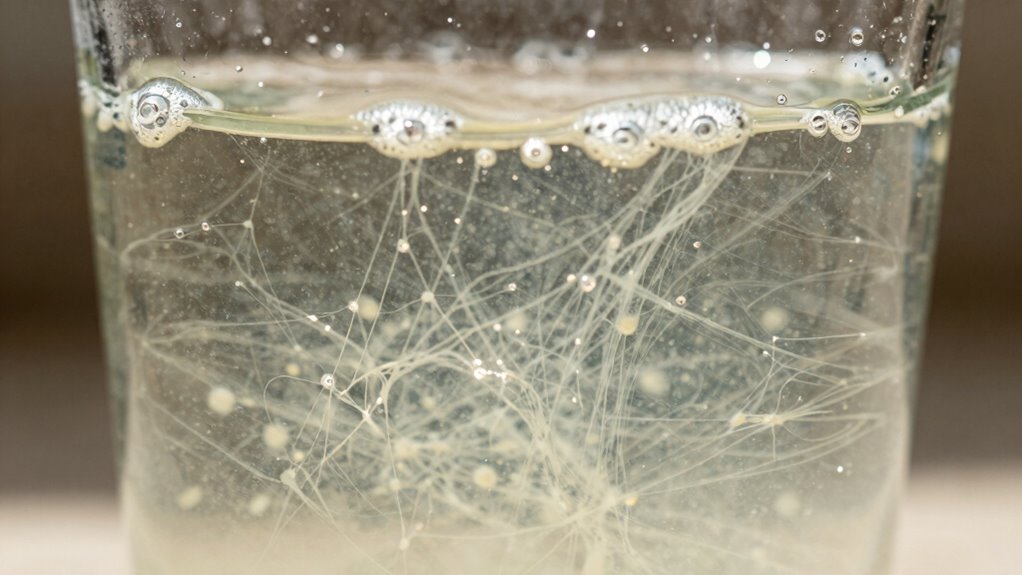
Dispersants improve cleaning in hard water by preventing mineral ions like calcium and magnesium from interfering with surfactants. These minerals can form insoluble deposits called scum, reducing detergent effectiveness. Dispersants bind to these ions, keeping them dissolved and preventing buildup. This allows surfactants to work efficiently, lifting dirt and oils without being blocked or deactivated. Additionally, monitoring market trends can help consumers choose the most effective detergents with dispersants. Here’s how dispersants help:
Dispersants prevent mineral buildup, enhancing detergent efficiency in hard water.
- Bind to calcium and magnesium, preventing scum formation.
- Keep mineral ions in solution, maintaining water softness.
- Enhance surfactant activity by reducing mineral interference.
- Improve overall cleaning performance, especially in hard water conditions.
Using dispersants ensures your detergent works better, saving you time and effort while protecting appliances from mineral deposits.
What Innovations Are Making Detergents More Effective and Eco-Friendly?

What innovations are making detergents more effective and eco-friendly? You’ll find that new surfactant formulations use plant-based ingredients, reducing reliance on petrochemicals. Enzymes have become more targeted, breaking down stains efficiently at lower temperatures, which saves energy. Cold-water detergents with advanced bleach activators work in lower temperatures, cutting energy use further. Microbial enzymes and biodegradable surfactants improve cleaning power while minimizing environmental impact. Additionally, nanotechnology enhances surfactant delivery, boosting efficiency and reducing quantities needed. Water-soluble packaging simplifies use and reduces waste. Researchers also develop enzyme-stabilized detergents that last longer and work better in hard water. Furthermore, incorporating sustainable ingredients helps reduce the ecological footprint of detergent production and use. These innovations help you clean more effectively while lowering your ecological footprint, making your laundry routine both powerful and sustainable.
Frequently Asked Questions
How Do Surfactants Interact With Different Fabric Types?
You’ll find that surfactants interact differently with various fabric types based on their fibers and textures. They penetrate and loosen dirt more effectively on natural fabrics like cotton and linen, thanks to their porous structure. On delicate fabrics like silk or wool, surfactants require gentler formulations to prevent damage. By adjusting surfactant types and concentrations, you guarantee ideal cleaning without harming your fabrics.
What Are the Environmental Impacts of Various Surfactants?
You might think surfactants are just cleaning warriors, but they also leave behind an environmental footprint. Anionic surfactants can harm aquatic life by disrupting ecosystems, while non-ionic types tend to be more biodegradable. When you use harsh surfactants, they can accumulate in water systems, causing toxicity and harming wildlife. Choosing eco-friendly options reduces these impacts, helping protect oceans, rivers, and the delicate balance of nature for future generations.
How Does pH Influence Detergent Efficiency Beyond Cleaning?
You’ll find that pH influences detergent efficiency beyond cleaning by affecting surfactant stability and performance. When the pH is within 7-11, surfactants work ideally, enhancing dirt removal and preventing reattachment. If the pH strays outside this range, surfactants may become less effective or degrade, reducing cleaning power. Maintaining proper pH ensures your detergent functions smoothly, improves stain removal, and minimizes residue or damage to fabrics and surfaces.
Can Surfactants Cause Skin Irritation or Allergies?
Yes, surfactants can cause skin irritation or allergies. You might notice redness, itching, or rash after contact, especially if you have sensitive skin or allergies. This happens because some surfactants strip natural oils or trigger immune responses. To protect yourself, wear gloves, rinse thoroughly, and choose detergents with milder, hypoallergenic ingredients. If irritation persists, consult a healthcare professional for advice.
How Are Biodegradable Surfactants Developed and Tested?
You develop biodegradable surfactants by selecting renewable raw materials like plant oils or sugars, ensuring they break down naturally in the environment. You test them through biodegradability assays, where you measure how quickly they decompose under controlled conditions. You also evaluate their efficacy in cleaning and their toxicity to aquatic life, making sure they perform well without harming ecosystems, aligning innovation with ecological responsibility.
Conclusion
While detergents may seem simple, they’re complex tools fighting grime and stains. You might think water alone can do the trick, but additives like surfactants and enzymes turn cleaning into a science. It’s a surprising contrast—hard water making things tougher, yet innovative ingredients making detergents smarter and greener. So, next time you reach for the bottle, remember it’s a delicate balance of chemistry and technology working behind the scenes to keep your world spotless.









